DIFFUSE CONTAMINATION
The multi-dimensional isotope approach can also be used for inorganic groundwater contaminants, i.e. nitrate and sulfate. Figure 3 illustrates the range of δ18O-NO3 and δ15N-NO3 values expected based on a variety of natural and anthropogenic nitrate inputs, and the fractionation effects that would alter these values. These observed differences in isotopic values can be used to distinguish between natural nitrogen loading (i.e. precipitation and soils), and nitrate runoff from other sources, including fertilizers, animal wastes, or septic systems. These isotopic ratios can also be used to assess the level and rate of denitrification that is occurring in a groundwater aquifer, which reduces nitrate concentration and thus reduces the impacts of nitrate toxicity for source waters.
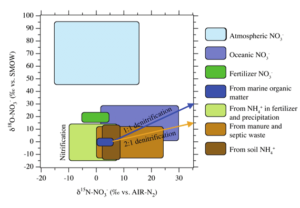
Figure 3: Typical δ18O-NO3 and δ15N-NO3 ranges for nitrate sources and the processes that alter these values (Kendall, 1998).
A similar approach can be used for the δ34S and δ18O isotopic values of mineral-bound and aqueous sulfate (Figure 4). These values can be used to look at the formation processes and conditions for sulfide ore deposition, assess the sources of sulfate to both marine and terrestrial environments, and examine the conditions and pathways for mineral oxidation (i.e. pyrite) that can adversely affect water quality through the production of acids.
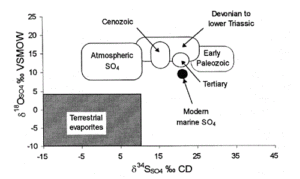
Figure 4: Range of δ18O-SO4 and δ34S-SO4 values for terrestrial and marine sulfates (Clark and Fritz, 1997).

